TECHNOLOGY
Fibrinogen Structure – Function
At the heart of Fibriant’s science and technology is Fibrinogen, a soluble blood protein that can rapidly be converted into an insoluble three-dimensional Fibrin network. In vivo this conversion takes place as a result of tissue injury and blood vessel damage that triggers the coagulation cascade which ultimately leads to the generation of Thrombin. Active Thrombin releases small peptides from Fibrinogen, activating sites by which the now Fibrin monomer molecules can interact with each other and assemble into Fibrin polymer strands, which subsequently become integrated into the insoluble three-dimensional Fibrin network.
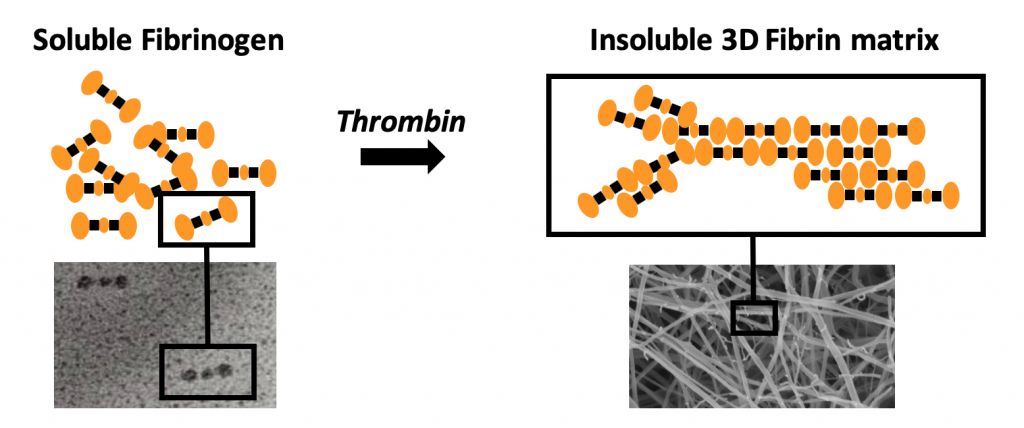
Schematic representation of Thrombin induced Fibrin formation. On the left a schematic model of Fibrinogen showing the two distal D domains and the central E domain (top) and an electron microscopic image (bottom) of individual Fibrinogen molecules before activation. On the right the “half staggered overlap” model of Fibrin polymerization (top) and an electron microscopic image of a 3D Fibrin network (bottom) are shown.
The Fibrin network is predominantly known for its important role in blood clotting where it provides a scaffold for platelets and other blood cells to form a mature hemostatic plug that seals the injury to the vessel wall, preventing further blood loss and initiating subsequent tissue healing. Recent scientific evidence demonstrates that the structure of the Fibrin network plays an important role in tissue repair, remodelling and regeneration by supporting cell migration and influencing cellular phenotype.
Fibrin and Fibrinogen also play a role in host-defense against invading pathogens and regulate processes such as inflammation and immune response. Elucidation of the exact mechanisms, involved in these non-hemostasis functions of Fibrin and Fibrinogen, could identify them as therapeutic targets to treat or prevent diseases in which one or more of these (patho) physiological processes play a role.
In addition to the many different functions, the Fibrinogen molecule also occurs in vivo in many different forms and shapes. Each of these Fibrinogen variants has unique functional properties and can form Fibrin matrices with different structural and functional characteristics. Several of these Fibrin polymers have specific properties which can be explored for unique healthcare applications. Most of the Fibrinogen variants occur at low quantities in blood therefore it is not feasible to obtain sufficient material of these individual variants by purification from human donor plasma.
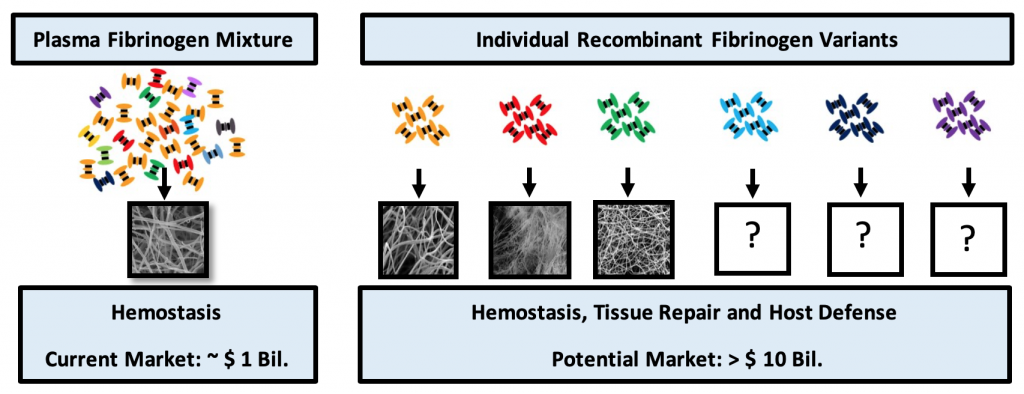
Fibrinogen isolated from plasma is a mixture of different Fibrinogen variants that form Fibrin with fixed structure and functional properties. Plasma Fibrinogen is currently only used in topical and injectable hemostat products. Individual variants of Fibrinogen, each with an unique structure -function profile, can only be produced by recombinant technology. The availability of these recombinant Fibrinogen variants not only enables development of customized hemostasis products but also creates opportunities in tissue repair and host defense applications.
Fibrin(ogen) Technology Platform
To take advantage of the unique functional properties of the individual Fibrinogen variants, Fibriant has developed a recombinant Fibrin(ogen) technology platform that is expected to create a large number of new, high-added value, customized Fibrinogen and Fibrin products with healthcare potential.
A robust up-stream process, using CHO cells, scalable, industry standard fed-batch conditions and stable clones, demonstrating high expression levels of fully functional individual variants of human Fibrinogen and active human Thrombin, has been established.
A down-stream process, based on a newly developed affinity resin, that allows purification of all different Fibrinogen variants, with high yield and purity, has been developed.
To characterize the different variants of Fibrinogen, Fibriant has developed several analytical methods that allow detailed structure-function analysis and can be used to determine quantity, purity, potency and stability.